gracielle
Registered
- Joined
- Jun 6, 2005
- Messages
- 4,089
- Likes
- 3,335

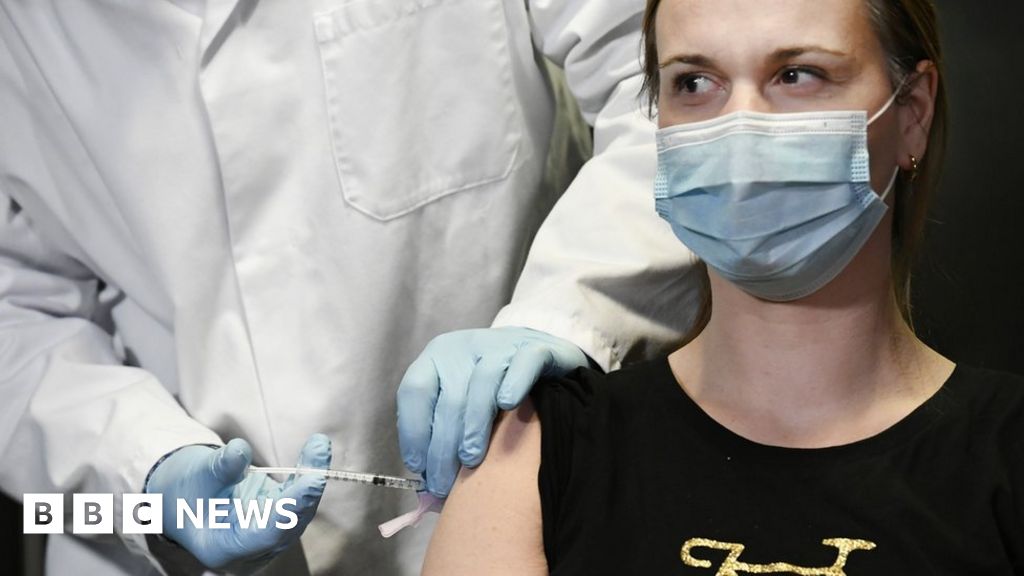
Soberana 02: Cuba prepara cien millones de dosis de la vacuna contra el coronavirus
Mientras la Organización Mundial de la Salud (OMS) mantiene una cruzada para denunciar el "
Soverana 02: Cuba prepares one hundred million doses of the coronavirus vaccine.
While the World Health Organization (WHO) maintains a crusade to denounce the "moral failure" of the concentration of the distribution of vaccines against Covid-19 among the richest countries, Cuba is moving towards phase 3 of its Soverana Vaccine 02 and is preparing to produce one hundred million doses.
"Cuba's strategy to commercialize the vaccine has a combination of humanity and impact on world health. We are not a multinational, where the financial objective is the number one reason. Our goal is to create more health," said the director general of the Finlay Vaccine Institute, Vicente Vérez Bencomo, when confirming the progress of the Cuban vaccine. For his part, the WHO representative on the island, José Moya, declared being "optimistic" in the advance of Soverana 02 and highlighted that "Cuba has been the first candidate from Latin America and the Caribbean to place its vaccine in clinical phase".
Soberana 02 began its expanded phase II clinical trial this week, which will extend to 900 people between 19 and 80 years old, according to the Granma newspaper, and an agreement has already been signed between the Finlay Institute and the Pasteur Institute of Iran to test phase 3.
Vérez Bencomo pointed out that the production of one hundred million doses will meet the needs of the country and also other interested nations such as Vietnam, Iran, Venezuela, Pakistan and India. In this sense, the vice president of Venezuela, Delcy Rodríguez, assured that Soverana 02 "will be the ALBA vaccine", referring to the alliance made up of Cuba, Venezuela, Bolivia and Nicaragua.
"The laboratories have already reserved almost all their production for the year and it is mainly the richest countries that have bought it, so these Cuban vaccines will be necessary," said Moya....
More than 30 years of sovereignty in health....
....Cuba, cornered by the US embargo imposed since 1962, has had to seek its own remedies. "In the 1980s they opted for biotechnology, discovering the first vaccine against meningococcus B", says Nils Graber, researcher in health anthropology at the University of Lausanne (Switzerland). "The objective was both to improve the national health system and to be an exporter," he adds, citing the delivery of treatments that Cuba has made to Latin America, Asia and Africa. The export of medical services, medicines, vaccines and medical personnel is currently Cuba's main source of income, with 6.3 billion dollars in 2018. In 2020, the island sent medical brigades to 40 countries to fight against the coronavirus. "The population would have perceived in a very surprising and regrettable way that Cuba would have had to import a Russian or a Chinese vaccine," observes the researcher.
Last edited: